or How to Use Cheap Butane
Jerry Adams with assistance from Roger Caffin
Appendix 1
or Why propane isn’t very useful in an upright canister stove if you want to use the entire canister at low temperatures
Given all the ideas in the previous three parts to this series, one has to ask what limitations restrict the use of upright canisters at below-freezing temperatures. It turns out there some significant restrictions. I took three different fuel mixtures and ran them through the spreadsheet used in the article The Effect of Cold on Gas Canisters by Stuart Robb and Roger Caffin to see what would happen to the propane concentration as the gas was used up. The results were a bit surprising.

The graph here is for canisters at -10 C (14 F). The X axis is how full the canister is: 100 = full canister (at left) and 0 = empty canister (at right). The Y axis on the right is pressure, and 1000 mBar is the nominal pressure at sea level.
The pink line shows how pure iso-butane would behave: the pressure stays constant of course as the composition does not change (it’s all iso-butane). The green line shows what happens if the canister starts with 20% propane: by about the 75% mark the boost from the propane is all gone. It has boiled off.
The pale blue line shows what would happen with a 50/50 blend of iso-butane and n-butane IF you were running it at very high altitude (low ambient presure). The iso-butane would evaporate slightly faster than the n-butane and the pressure would keep slowly dropping. The dark blue line shows what you would get with a propane/iso-butane/n-butane blend: the pressure starts higher but still cascades down to a similar very low end-point. However, and this is important, that would only work at extreme altitude. The pressure at sea level is 1000 mBar, so in practical terms you would never get any gas out of the iso/n canister at sea level at -10 C, and the canister with some propane would terminally die when it was half-empty.
A technical recapitulation: if the pressure is greater than 1 atmosphere or 1000 mBar (the Y axis) then there will be some pressure to the stove and it will work at sea level. Actually, it needs to be a little higher, say 1100 or 1200 mBar, to provide enough pressure for a good burn (corresponding to boiling a pint of water in less than 6 minutes with my stove). If you are at 3,000 m where the ambient pressure is lower, then a slightly lower pressure is enough to get the stove to work – but the ambient temperature may be a bit colder.
Therefore, to use a canister at cold temperature, you want as little N-butane as possible. That really is the crunch point. Whether you can rely on a label which says ‘iso-butane’ – that’s a different question.
In the petroleum refining process, it’s easy to separate propane because the boiling temperature is so much lower than butane, so propane is cheap and widely available. It’s more difficult (= expensive) to separate iso-butane from n-butane because their boiling temperatures are close together. It’s much easier to make a canister fuel that works when the canister is new at low temperatures by just adding back some propane than removing the N-butane. The fact that the benefit does not last is just tough luck.
Could the ‘blends’ we see on the shelves just be what the manufacturers can get out of their available feedstocks at the lowest possible price? Is a blend of propane/iso-butane/n-butane be just what the incoming gas was, without any treatment? And could it vary a bit from shipment to shipment? One wonders.
But note that this is all for upright canister stoves – remote inverted canister stoves don’t have this problem as by design there is no preferential evaporation going on. Adding propane will very helpfully lower the usable temperature for the entire canister. N-butane is okay if balanced by a good amount of propane, but you’re still better off with less n-butane. The Cold Canisters article mentioned above gives results for a number of common fuel blends: use the values for when the canister is full.
Appendix 2
or How propane depletion is barely changed at cold temperatures
Let’s look at this from a different direction: how does the temperature affect the depletion of propane from an upright canister? Again, I used the spreadsheet from the article The Effect of Cold on Gas Canisters, this time to see how much propane is left when you start with canister containing a 20% propane / 80% isobutane mixture and use it at different temperatures.
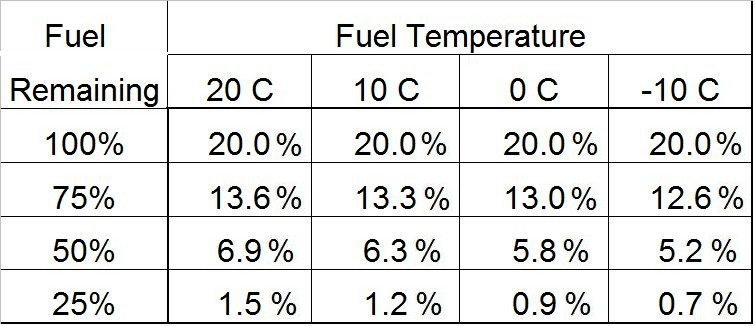
The amount of fuel left in the canister is given at the left. The temperature is the top line of numbers. The rest of the numbers are the percentage of propane left for the given conditions.
The startling result is that changing the temperature from +20 c to -10 C has very little effect on the composition by the time you get down to a half-empty (50%) canister. Running at 20 C there is only 6.9% propane in the liquid remaining in a half-empty canister; running at -10 C there is only 5.2% propane left. This very small difference corresponds to about 1 C (2 F) difference in the boiling point for that canister.
It is commonly said that there is more propane depletion when you run at colder temperatures. This is so because the propane is still (as it were) boiling away but the iso-butane component is not. Well, the data in the table is consistent with this, but the effect is minor. I think it’s better to just accept that propane is going to be seriously depleted regardless of the temperature you run it at, IF you used the canister upright. And keeping the canister warm does not win you much towards the end either.
Once again, none of this applies to the use of an inverted canister stove. There is a reason they exist, after all.




