Hi Dan
I would think that if there was CO/gas vapor coming of it would ignite.
Life is never that simple. There is always a barrier between to be overcome before two atoms can join or burn. Those little flames may not be enough.
For a start, the oxygen molecule O2 has to be broken in half (dissociation) into 2-off free oxygen atoms, and that takes energy. Then two atoms have to overcome the mutual electrostatic repulsion due to the surrounding electron clouds, before they can be close enough to share electrons. That requires a high atom velocity, aka temperature, for them to collide.
Reminds me a little of courting . . .
With a normal flame, it is the energy given off by the current ‘burning’ atoms which provides the free energy for the dissociation of the next set of molecules.
When you look at a flame you normally see two different blue colours: light blue and darker blue. These represent different chemical processes. The first set involves free hydrogen burning, as it can be broken off from propane and butane molecules more easily. Only later on at a higher temperature are the carbon-carbon bonds broken to allow carbon-oxygen bonds to form.
So how do you get a flame in the first place? If you are using a piezo igniter, it is the glowing flint spark which starts the process. The glowing spark comes from mechanical action, steel on flint. But if there is not enough fuel in the gas mix, or if there is too much fuel in the gas mix, the energy barrier can be too high and the spark dies. That is why piezos often do not work at high altitude: the mix is ‘wrong’. Matches? Mechanical friction between the match head and the side of the match box.
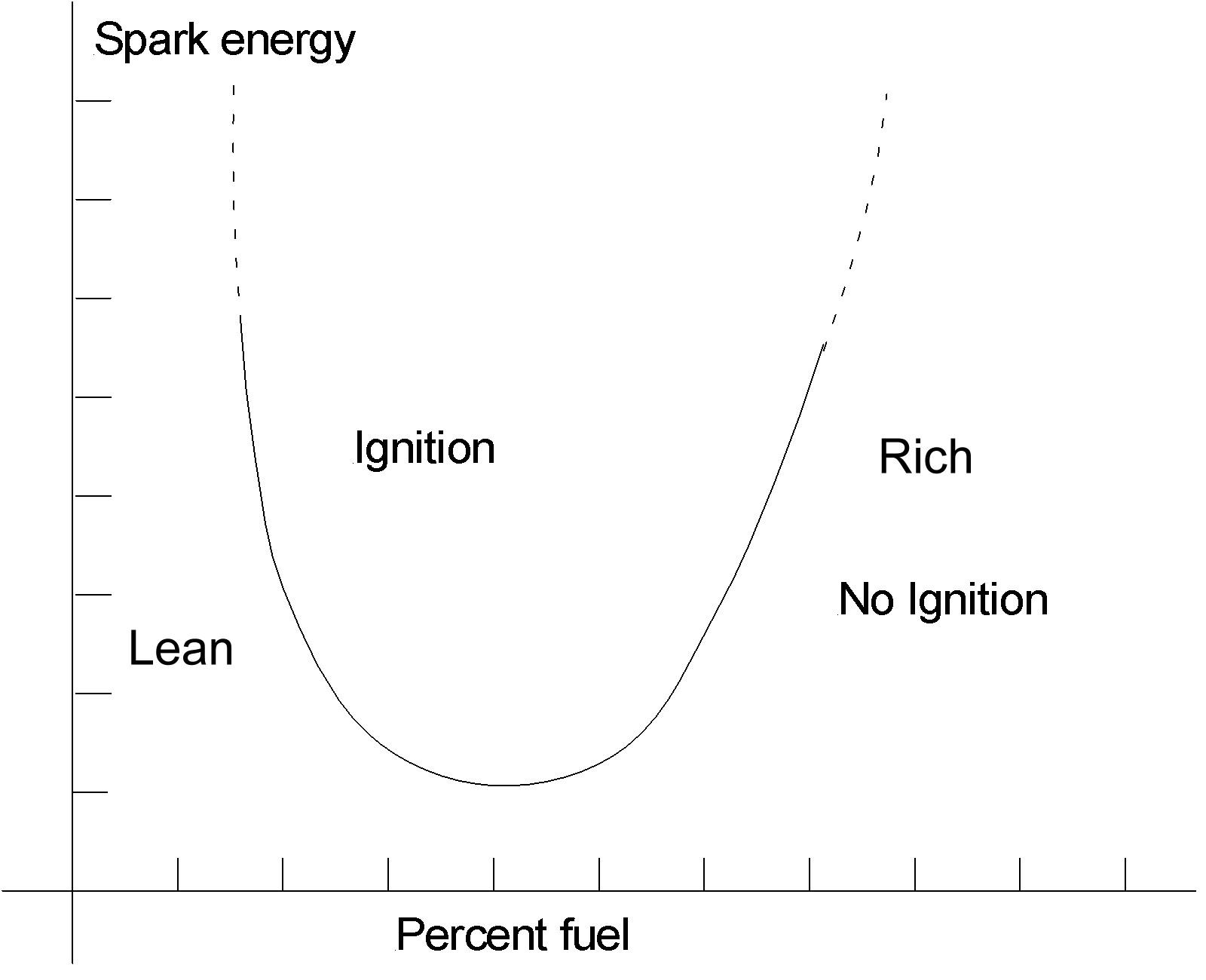
The diagram here illustrates the effect of the fuel/air ration on the ease of ignition.
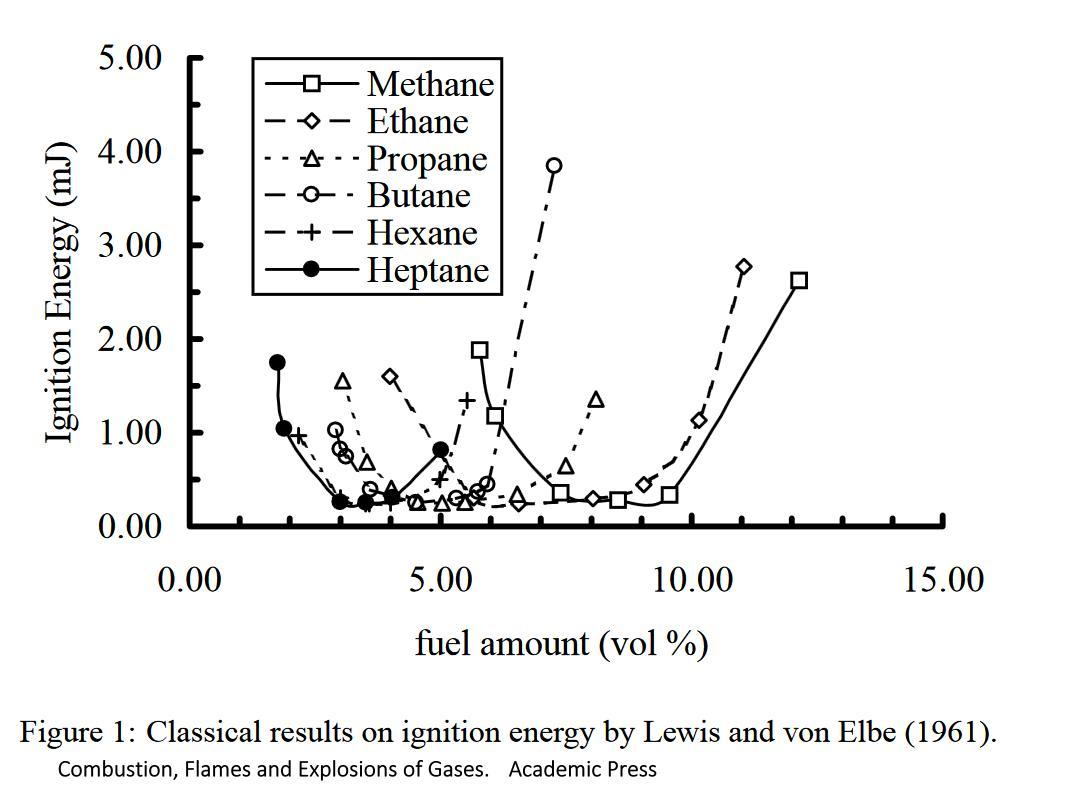
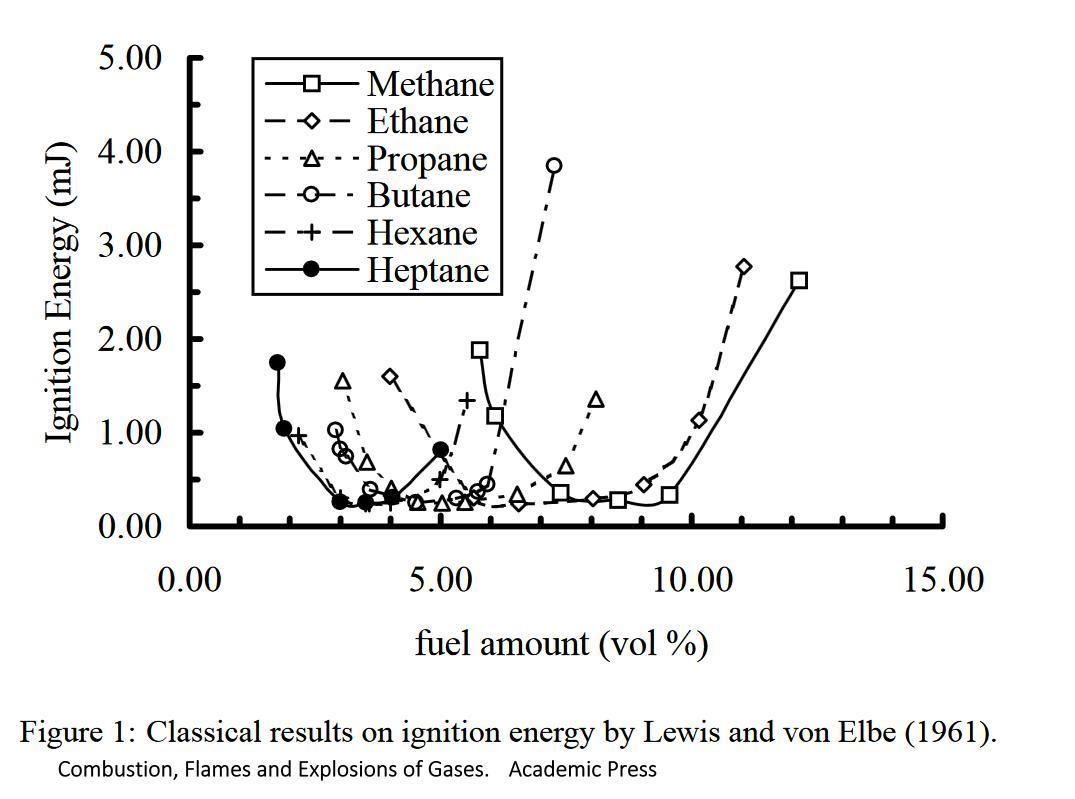
Too little fuel or too much fuel and the energy required goes right up. I do have a simpler diagram, but I cannot find it right now.
”The chemistry of a flame is fascinating . . .”
Cheers