Oliveir A, Well, there are calculations and other calculations. I am not sure what they use on canisters. 70/30 is in percent, soo, simply multiplying thru with 30% on an 220gm canister, yields 66gm of propane in a can. In chemistry, they add up molecular weight (moles) at standard pressure/temperature(STP) and multiply through with 0.3 to get the total amount. There are other PERCENTAGES possible (say ratio of pressures) depending on what you are looking at, but I suspect that the first calculation is close enough for what you want.
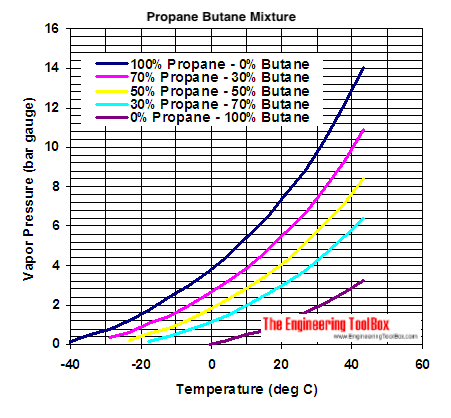
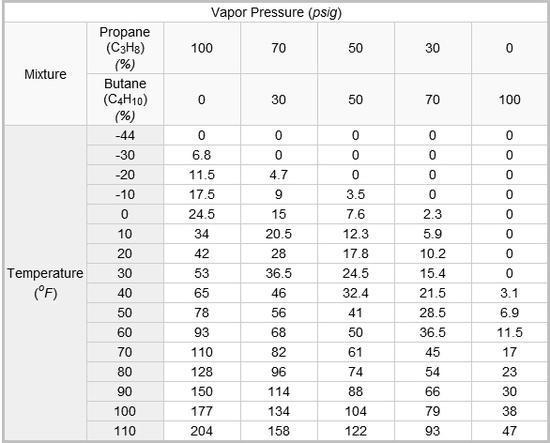
I’m not sure 100% propane is possible except at frigid temps (ie, <32F or 0C.) Pressure increases with temp increases, or, the corollary, pressure decreases with cooling. Generally the boiling point of propane is much lower than the butane family (n-butane, isobutane.) So, keeping the temp at say -43F (the boiling point of propane) you could load it up with 100% propane with no problems. Buy, of course, it would not power a stove at that temp, either. Most canister fuels started with using waste butane gas from refineries. This was a mix of all sorts of gasses, actually, but generally n-butane & isobutane were the primary ingredients. By isolating isobutane, more “heat” could be packed into the same can,, ie more burnable molecules at less pressure. Adding propane adds more heat, but also the corresponding pressure increases, since the propane fraction in the can is increased. So, the same specifications on a can will allow a 75/25 mix of isobutane/propane. 70/30 is close to the maximum. This is about the limit for cans with a safety margin. Why? Because more propane increases the pressure inside the can. Adding propane increases the cold weather performance, also. Propane will tend to fractionate (a type of distillation) into lower percentages when in use, since it will boil (increasing pressure) at a lower temp than butane. You start running into gas laws, and other calculations, of course. As an example, there are stories of half full cans that will not burn when connected to a stove at low temps. The propane has been previously burned off when winter hiking/camping, hence little is left to power a “topper” stove.
So it is a curve of temp/pressure that will determine when you can use increasingly pure propane. As a rule of thumb, I would use no more than a 50/50 mix at any normal winter hiking temp. This will give you some margin of safety at 32F/0C, less with any increase in temp (say carrying a can in a car to/from a trail head.) Cans will only contain so much. pressure Letting them warm up to say 81F/27C, could potentially exceed the safety standard on a can, and have it explode in your backpack I believe Roger C examined this in much more detail than I have. Perhaps he will chime in. Unfortunatly my chemistry is 50 years old and I don’t remember the exact calculations involved.
It doesn’t really matter if there is more room in a can. DO NOT FILL IT UP. The space provides a buffer between liquid (propane/butane, etc) and gas. Common acetylene cylinders use a butane mix to absorb the more reactive acetylene molecules, thus lowering the pressure in a tank. They still only add just so much and leave about 15-20% empty space. Think of boiling water in a sealed container. High pressure steam will explode a container IFF there is no room for thermal expansion! Even water bottles should not be filled to 100% when hiking, they can produce leaks because of the temp/pressure. (Freezing a beer in a closed container will usually result in a mess.)
Anyway, I would not use 100% propane in a standard canister. Again, 60/40 or so is about the most I would risk. I believe an old, obsolete canister design based on a smaller cylinder was used unsuccessfully 20-30 years ago at 60/40. But this was designed around the shortfalls of a standard canister.