Cool! A topic relevant to what I do! So… time for a lengthy response. Trust me, I’m a water engineer :)
Before I get into the technical stuff, just a few comments on that desal filter. This is MY OPINION based on the knowledge I have. To each his own:
- A few liters per hour is SO SLOW and not practical. I don’t want to even think about having to pump water for an hour.
- As Jerry mentioned, reverse osmosis (RO) takes electricity, and more generally A LOT of energy (in terms of pressure). That’s of course scale-able depending on what’s actually in your water and how fast your trying to produce it. I’m working on a design for a water treatment plant (WTP) using RO technology. The pumps are… about 1000 hp and $500,000+ for each! There’s also a lot of other side process (which aren’t really an issue with a small pump like that).
- I guess you step on it to hold it down, and then extend the lever up, which helps with leverage. Still seems like more effort than I care to exert.
- A Sawyer filter is cheaper, MUCH lighter, and filters out most of what you need it to (see images below for reference). Drops for chemical treatment or a UV pen are the icing on the cake, and similar to the disinfection stage at a typical WTP. I’ve never used either of those while backpacking and I’m alive. But for the weight, and depending on your water source, filtration PLUS disinfection is best to avoid diarrhea (or something else) due to bacteria/viruses.
- CONS: I’ve never tried it personally, but a Sawyer filter won’t be effective on seawater or salty water since it’s not designed to remove ions.
- They have other reverse osmosis (RO) units that work for things like boats. That’s a more practical application.
- The filter only seems useful to a person backpacking along a saltwater body with no access to anything else. It’s pointless in any other backpacking situation in my mind, unless you plan on filtering your own urine or from a virus/bacteria ridden lagoon. That just sounds gross. I’d rather have dry mouth for a while.
- But, $60 is interesting just to see how it works. Not a great option for backpacking in my mind.
- Lastly, “is said to meet WHO standards.” LOL. Just…. LOL. I love when someone tells me things I can believe. I’d be a little more reassured if it said “meet” instead of “said to meet”. I’m sure some can agree :)
Ok… now for the poorly-written technical stuff:
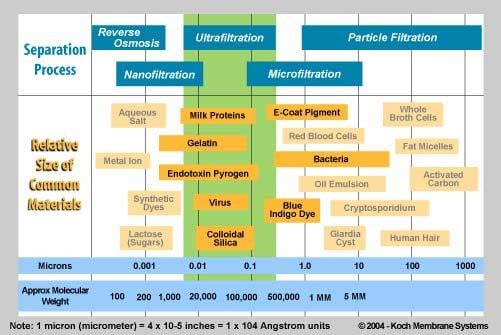
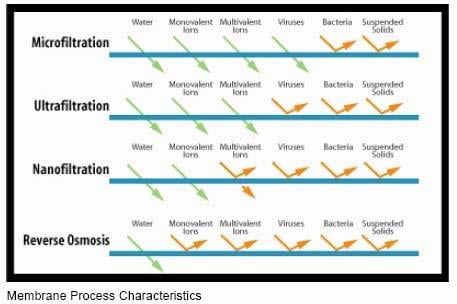
There’s filtration, and then there’s RO. RO is technically not filtration. You do see it in a lot of coastal communities, or places where the only available water source is deep underground (ultimately having a lot of dissolved ions due to minerals leaching into the water). There are also different levels of filtration. By order of what process removes the most constituents from water from less to more, it’s microfiltration (MF)<ultrafiltration (UF)<nanofiltration (NF). A Sawyer filter is considered an ultrafilter per their site (0.1 to 0.02 microns). Per the two figures below, it’s plenty good IMO for removing viruses and bacteria (what you really care about).


However, per the above, if you are backpacking in places with high mineral deposits and places that have heavy metals regularly, maybe explore other options? Drinking water with some Barium in it during a weekend trip isn’t going to kill you. But drinking it every weekend? Probably not the best way to stay cancer free. It’s not much of an issue in places with glacial-fed rivers, but more so desert areas.
RO removes more than the three types of filtration I mention above, but RO membranes technically don’t “filter” anything out in terms of the science and language, really. In practice… sure… it removes microscopic stuff from water to be non-technical. It uses a semipermeable membrane (probably similar to the way a membrane-type outdoor fabric lets air through and “breathes” but keeps water out, if you will) to act as the “filter” and desalinate <span style=”background-color: transparent; color: #333333; display: inline; float: none; font-family: Georgia," times new roman","bitstream charter",times,serif; font-size: 16px; font-style: normal; font-variant: normal; font-weight: 400; letter-spacing: normal; line-height: 24px; orphans: 2; text-align: left; text-decoration: none; text-indent: 0px; text-transform: none; -webkit-text-stroke-width: 0px; white-space: normal; word-spacing: 0px; word-wrap: break-word;”>water </span>(or, remove salts).
Any solution naturally “wants” to move from higher concentration to lower. So if you put, let’s say, saltwater in one bucket and tap water in another, separated by a pipe with some semipermeable membrane, the tap water will move to the other side and they will eventually reach an equilibrium. The two buckets will have the same water quality, more or less, since water molecules can pass through the membrane but the salts cannot. RO works by applying pressure to the saltwater side (or high concentration side) so that water (H2O molecules to be super general) goes through the membrane, but other undesirable constituents (dissolved solids, viruses, bacteria, etc.) don’t pass through. This produces a permeate (“filtered” water with low concentrations of undesirable constituents… or “bad stuff”, and concentrate (the waste stream of water with a high concentration of “bad stuff”.
RO permeate in itself is technically drinkable, but it has very little minerals. So it’s not good to drink for a sustained period of time, because it will more or less leach minerals from your body. It’s just like how your body needs electrolytes (salts) rather than just water. So, utilities usually blend the permeate with pre-filtered water (which hasn’t gone through the RO process) to make a more stable water product. Permeate as is has a pH of 5 or 6, usually, and will corrode piping!
RO is FAR SUPERIOR in the ability to remove “bad stuff”, but it’s also not necessary in every application. I think backpacking is one of them. Now if they could make a filter the size of a Sawyer, but with a nanofilter, that’d be your ideal one-stop-shop. Who knows… maybe they have them for backpacking already. It’d remove viruses and bacteria, and your multivalent ions (+/- 2 charge on the ion or more, to get into the chemistry here). We ultimately need certain amounts of multivalent ions like calcium, magnesium, and even metals like iron, but too much of them isn’t good either. And that’s why the DEP sets concentration limits on all of these things. There’s a lot more that goes into treating our water than we realize when we stand there and watch it come out of the tap.
To end… after typing all of this, I probably could have just posted a link to something someone already wrote out (probably much more eloquently). Oh well. And don’t quote me on this stuff. I’m not a licensed engineer yet, nd I got lazy towards the end and didn’t want to fully proofread :)