The link from Greg seems to be reporting all relative pressures for me., as does the Weatherbug site. Denver should be around 25 inches on a normal day, so when they're reporting around 30 inches, I know it's not absolute that they're reporting.
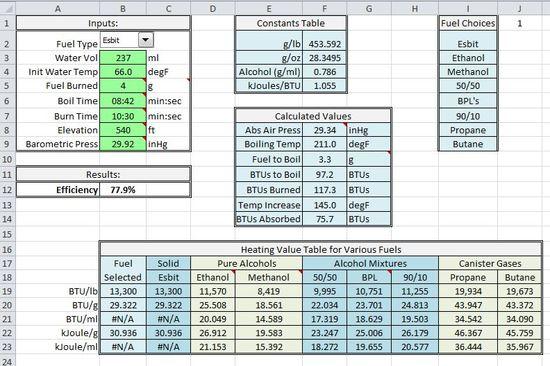
The conversion calculator Galen linked to, can take the relative pressure and convert it to absolute, which we could then use instead of a compensated elevation, but that's the very thing we're trying to compile into a single process with spreadsheet formulas. I'm using the Antoine equation to do this. Probably not the best, but at the time, there was no input from anyone on this matter, so I winged (wung?) it. It's close enough when used in this manner, that I believe any discrepancies are below one tenth of one degree for boil temps, which would effect the efficiency rating even less than that.
I think maybe the newer electronic consumer level barometers would be able to do this, but I have no idea what they cost, or how viable it would be for the average tester, I can't speak to that. But yes, a normal home barometer with a dial has a range of around 28-31, so there is no way to get absolute pressures with them. You guys can do what you want with advancing this project, but as I've repeatedly stated, MY goal is to keep it simple enough that "someone's mother" could perform these tests without any specialty gear. Since relative barometric readings are what's commonly reported to the general public, that's what I'll be using. The only thing I might change is the formulas for converting them, when/if something better arises.
Sometimes it's the "pro", military-grade model that gets stripped down for easier mass consumer use, sometimes it's the simple everyday product that gets beefed up for more "professional" and industrial use. This project is currently following the latter IMO.