I posted a review of the MSR Hyperflow in the reader reviews section of BPL. I know there has been some interest expressed here thus the pointer.
Topic
MSR HyperFlow filter
Become a member to post in the forums.
- This topic is empty.
I have heard that a major problem with this pump is that it can break when frozen. Has anyone else heard of this. The person that told me this told me that the pump will still act like it is working when, in fact, it is not truly filtering water. If this was true I guess you could keep it inside your sleeping bag on a freezing night.Not meaning to spread bad rumors of this isn't true.
Josh
> it can break when frozen.
MSR specifically and clearly warn that allowing the filter tubes to freeze will damage them to the point of failure.
The rest of what you said is also correct.
We will publish a review of this item when we have had enough field time with it.
Cheers
Surely freezing water inside any filter would damage it? It makes sense to me anyway since water expands when it freezes it would surely damage the filter element.
If the filter element got damaged, how would you know? I only know of one filter where you can test/check it in the field: The First Need. Are there others?
This is done by putting 2 drops of dark food coloring in 1 cup of water. Pump it through and if water comes out totally clear, the element is still good. If any slight faintness of colored water—then bad :(
-Barry
Having used the Sawyer inline filter, which uses the same hollow fiber technology as the Hyperflow, the dye test should not work.
If there is active charcoal in the filter, it would remove the dye color, which would show that the filter is working.
In the case of using the Sawyer inline filter, I have put green/brown water through it and it came out greenish.
The Sawyer rep that I spoke to at REI says that things at the molecular level, such as electrolyte powers, will pass through the hollow tubes/fibers, but larger things like bacteria will not.
Unfortunately, the negative to this technology is that you are working completely on faith.
If it freezes and is damaged, I don't know how you could tell unless large particles were passing through to the clean water bottle that you are drinking out of.
If the particles are coming through, something is really screwed up inside of it.
-Tony
Barry:
I am a new owner of the First Need purifier. I wonder if it's really the filter/purifier components or simply the carbon component that's removing the color?
Everyone:

Curious, for those who got a Brita filter and some food coloring, might you be agreeable to doing a quick test for us? We know that Brita filter contains a carbon element to improve water clarity and taste but it is basically useless against biological contaminants. So if your Brita can turn dyed water into clear water — then we'll know that the First Need test is fatally flawed.
Tony – Just blow or pump air through the filter with the output end under water. If any of the hollow fiber tubes are compromised there will be localized big bubbles rather than dispersed fine bubbles.
Ben – It is only the activated carbon that removes the coloring. This is a common grade school science experiment. There is no need to gum up a Brita.
FIRST: Filters should never be used in freezing temps. That's what they make UV light purifiers & chlorine dioxide tablets for.
SECOND: If you have used a filter and suspect temps will get below freezing at night be sure to empty the filter of water as much as possible, even to the point of disassembly. Then reassemble, put it in a Ziploc or small drybag and keep it in your sleeping bag overnight.
Eric
Richard:
Food coloring will hardly gum up anything — although yeah, what you said pretty much confirms my suspicion that the First Need purifier "test" is nothing more than a 'snake oil' demonstration to impress the masses. Not saying the purifier doesn't work — just saying the "color dye test" is near worthless.
“..might you be agreeable to doing a quick test for us? We know that Brita filter contains a carbon element to improve water clarity and taste but it is basically useless against biological contaminants. So if your Brita can turn dyed water into clear water — then we'll know that the First Need test is fatally flawed.”
Hi Ben,
I think that is an excellent idea. I pulled out my Brita pitcher. I read my filter box and they use an ‘activated carbon’ filter element. I put 2 drops of food coloring in a cup of water… pour it into my pitcher… wait… wait… the water slowly trickles out, and it’s totally blue! The filter element didn’t filter the blue at all. Because of this, I don’t think the “"color dye test" is near worthless”, but very valuable! It really shows the First Need filter is working.
Thus it appears the First Need does an excellent job at filtering organic molecules. I don’t quite understand what’s in their proprietary ‘canister matrix.’ But it seems regular activated carbon filters don’t do as well as the First Need.
I also have a ceramic filter that I tried the dye test on and it let some of the blue through so it did better than the activated carbon filter.
So again, I beg the question, is the First Need the only filter out there that you can tell it’s still working in the field? Filter failure is just not from freezing but also from dropping.
The suggestion by Richard to blow through the Hyper Flow and check the air bubble formations seems way too variable to really determine filter status.
-Barry
Thank you for doing the test, Barry!!!
So a carbon element cannot "block" the blue dye by itself. This provides more confidence that the dye test will work only if the First Need's filter component is working properly.
Perhaps I was too hasty in my assumption. Just goes to show that even "grade school science experiment" is worth doing and confirming! I must admit though that I am pleasantly surprised by your Brita test because I felt "for sure" it was the carbon doing the work! After all, we know the Sawyer is also a purifer but it lacks a carbon element and it does not turn colored water into clear water…
Barry,
Your activated carbon analysis reminds me of an experiment in which someone cuts the legs off a frog, yells at it, and when it no longer jumps, concludes: "When you cut the legs off of a frog it goes deaf" Brita recommends that their pitcher filters, #3551, should be replaced every 40 gallons (about 2 months for the average family). How many gallons were previously processed by this activated carbon filter? For a comprehensive scientific explanation, of how activated carbon removes blue dye, order a reprint of http://www.springerlink.com/content/n34w57lt46112w70/
Have you tried the air bubble procedure I recommended to test for hollow fiber rupture before concluding it is too variable to determine filter status? I suggest that you look at the pictures in the MSR Hyperflow filter test guide part 3.
Hello Richard,
“How many gallons were previously processed by this activated carbon filter?”
Well, the answer is zero. I used a brand new filter. And it’s not limited to Brita. I had the EXACT same results with the Katadyn Hiker Pro and their activated carbon filter—and it was brand new. I even changed dyes and used a supermarket red dye; same results; First Need works, the other filters don’t. So it seems activated carbon filters just don’t filter out dye.
Also, as of this posting time, I couldn’t get your link to work.
“Have you tried the air bubble procedure I recommended to test for hollow fiber rupture before concluding it is too variable to determine filter status? I suggest that you look at the pictures in the MSR Hyperflow filter test guide part 3.”
Pictures are worth a thousand words. Where may I look at this test guide picture w/o buying a Hyperflow? It just seems amazing how fine bubbles become a big bubble just by compromising a hollow fiber tube. I would theorize that several tubes would have to go bad before a big bubble was to be formed. And by then it’s too late.
Thanx for your insight,
-Barry
Barry,
Standard high school experiment showing how dye is removed from water
MSR Hyperflow Hollow Fiber Field Integrity Check

(EDIT – See P. 2 discussion. My results discussed below were flawed and blue dye was NOT actually filtered out).
I know this is an old thread but I was searching this subject recently and someone else might be able to add to my experience.
I own two Sawyer filter. One is new, one a year old. I took the old filter out on several subfreezing nights. I kept it in a ziploc bag next to my pad under my quilt. I decided to test it after a trip where temps fell to 22 degrees F using infamous blue dye test. That night my water bottle (in same location) didn't freeze but I thought the quilt came off the filter during the night.
I used McCormick food coloring which contains a combination of Blue Dye 1 and Red Dye 40. It failed the test. I wasn't sure if this is a problem because I don't know the micron size of this dye.
So I decided to buy a new Sawyer filter. Tested side-by-side with the same dye solution, the old filter ran blue and the new filter ran clear. At this point my old filter has bigger pores or a crack inside.
I then tested McCormick Yellow Dye No. 5. Both new and old filters ran yellow, so this is a smaller molecule (or a solute) than blue dye. I then tested both dyes on a coffee filter. The yellow dye migrated further, indicating it is a smaller molecule, and the blue a larger molecule.
Bottom line, I'll hike with my new filter and keep it in the foot of my bag at night where it can't get uncovered.
I'm pretty convinced the carbon element is the main factor at work here. Dyes are organic compounds….run them through an activated carbon filter and they're going to stay there. I doubt it has anything to do with the filter's micron rating.
I too tried this with an old and new sawyer filter….the old one had been exposed to borderline freezing temps so I purchased a new one. The old one ran blue with the dye test…..but so did the new one. Throw in a platypus carbon element I carry around when I'm filtering near populated or agricultural areas, and both filters ran clear.
Not all dyes are organic compounds. Some, such as common rust, is not organic yet it can dye cotton stuff fairly well.
Jama, I'm surprised to hear the results of your blue dye test. The pores in the Sawyer filter are a tenth of a micron, which (you might know) is far larger than any dye molecule. The term "dye" is usually used for solutes, and the term "pigment" is usually used for suspended particles. Food coloring is typically a dye solution, whereas white paint, which contains titanium dioxide particles, is a suspension of pigment.
I can only imagine two explanations for retention of dye by a filter with 0.1 micron pores: the colored test solution is a pigment suspension (not a dye) or it is a dye solution and the dye molecules have some affinity for one of the materials inside the filter (the polyethersulfone tubes or some other component), and this affinity changes as the filter ages.
I'm going to look for the McCormick Blue 1 Red 40 mixture and look at it under the microscope to see if there are particles. You found that McCormick Blue/Red dye at the grocery store, I assume?
Yes, McCormick food coloring from grocery store. This is a 1 oz bottle (rather than the group of 4 mini squeeze bottles). The labelled contents are …
Water, propylene, glycol, FD&C Blue 1, FD&C Red 40 and propylparaben (as preservative)
Maybe the blue dye binds to another molecule among those bottled contents and that larger molecule isn't passing through. I don't know the molecular size of this dye solution, but maybe McCormick will tell us.
I accept that the blue dye experiment may prove nothing related to water safety …but it would be great if there were some sort of .1+ micron test kit. One of you DIY-ers should come up with that product. I'd buy it.
Okay, I asked McCormick if they can shed any light on the molecular structure of they blue dye solution.
I think someone suggested the dye might bind to new material inside the new filter but the dye did backwash out again.
Very interesting
I've had my Sawyer Squeeze in freezing temps a couple times, but I'm pretty sure the filter didn't freeze because there were water drops on it. I'll have to do the blue test anyway.
So, I assume you put a couple drops of blue in a liter of water (or whatever), filter it, if the filter is good the filtered water will be clear?
FD&C Red 40 is 2-naphthalenesulfonic acid:

It is a small and simple organic molecule. A typical protein is far larger than this, and it takes trillions of proteins to make a mycobacterial cell about the size of the pores in a Sawyer filter.
FD&C Blue 1 is sometimes called erioglaucine:
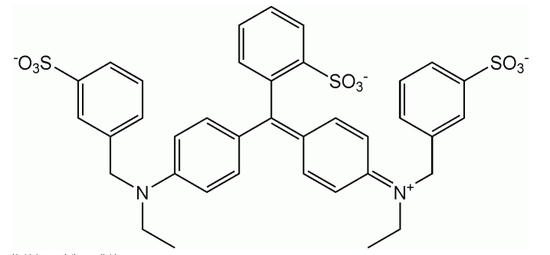
It is also a small, simple molecule. The tinyness of these, relative to the pores in the Sawyer filter, cannot be overstated. Trillions of these could be lined up end-to-end across one pore in a Sawyer filter.
Jama, your results stump me. Is the dye viscous (syrupy)? I can't fathom why these dyes would be retained by a filter with 0.1 micron pores and then backwashed out again. Hopefully we will learn something interesting from this. It would be very handy to have a simple dye test to confirm the integrity of these filters.
How did your filter freeze? Was the entire filter full of water?
With the Squeeze, you can shake some of the water out. I assume this is the water on the outside of the tubes, but the tubes are too small so you can't shake the water out of them.
I assume in your case, there was water inside the tubes, and also outside of it, because it's not convenient to shake this out with an in-line filter.
From wikipedia, formula for blue #1 is C37H34N2Na2O9S3. Maybe diameter of molecule is big enough to be trapped by filter?
wikipedia says maximum absorbtion is 628 nm. Does that mean the molecule is 628 nm? = 0.6 micron? the filter should easily filter that.
Become a member to post in the forums.




